Genome-scale Screening Identifies Key Genes for Pluripotency Exit of Rat Embryonic Stem Cells (ESCs)
Schematic diagram of the process in which Thop1 affects the pluripotency exit of rat ESCs by affecting the phosphorylation of ERK1/2
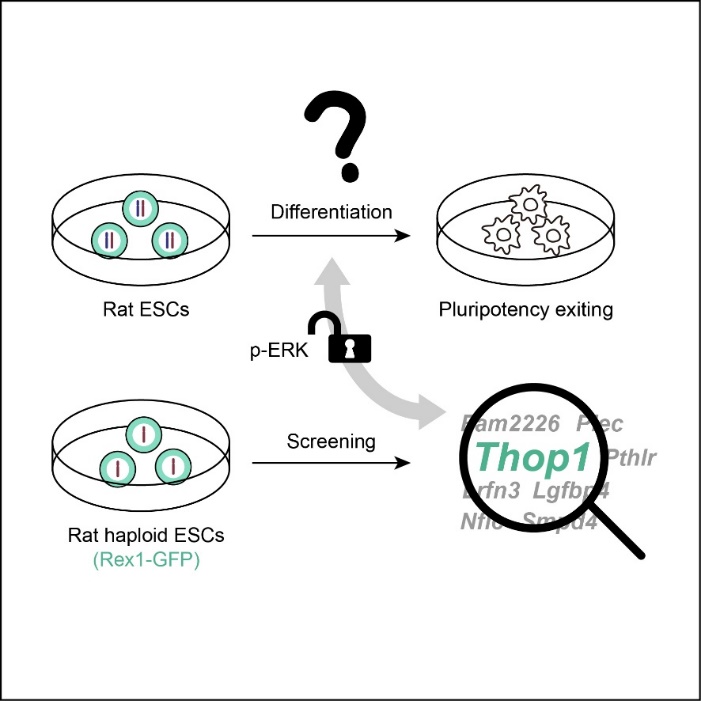
Recently, the research group led by Researcher Shuai Ling from the State Key Laboratory of Medicinal Chemical Biology of Nankai University has screened the rat haploid ESCs (haESCs) carrying a Rex1-GFP reporter and found that knocking out Thop1 can inhibit the phosphorylation of ERK1/2 and thus retard the pluripotency exit (random differentiation) of rat ESCs. This study not only proves that haploid genetic screening system shows great advantages in studying the mechanism of the self-renewal or pluripotency of rat ESCs, but also provides a new theoretical basis for the mechanism of the pluripotency maintenance and exit.
In this work, the research group led by Shuai Ling conducted genome-wide mutations of haESCs using piggyBac transposon, and obtained a genome-wide high-throughput mutation library. Subsequently, the target mutation library of delayed differentiation of rat ESCs was identified by random differentiation under the guidance of Rex1-GFP reporter. Finally, High-throughput sequencing analysis reveals numerous insertions related to various pathways affecting random differentiation.
On this basis, through gene editing and other experiments, the researchers confirmed that the representative gene Thop1 can regulate the pluripotency exit of rat ESCs by affecting the phosphorylation of ERK1/2. Under the culture condition of random differentiation, knockout of Thop1 can significantly retard the differentiation of rat ESCs, and the phosphorylation level of ERK1/2 in rat ESCs is significantly lower than that of wild type. Otherwise, under the normal stem cell culture conditions, overexpression of Thop1 can promote the differentiation of rat ESCs and increase the phosphorylation level of ERK1/2.
On March 11, the paper introducing the research results was published online in the journal Cell Proliferation. Xu Mei and Zhao Yiding, doctorial candidates from School of Pharmacy, Nankai University, and Dr. Zhang Wenhao from the State Key Laboratory of Medicinal Chemical Biology, Nankai University are co-first authors. Dr. Gao Qian and Researcher Shuai Ling from the State Key Laboratory of Medicinal Chemical Biology, Nankai University are co-corresponding authors. The research was funded by the National Key Research and Development Program of China, the National Natural Science Foundation of China, and the Strategic Collaborative Research Program of the Ferring Institute of Reproductive Medicine, etc.
Link to: https://onlinelibrary.wiley.com/doi/full/10.1111/cpr.13209
(Edited and translated by Nankai News Team)









