New Breakthrough in Hydrogen Energy! NKU Team’s International Cooperation Results Published in Nature
On the evening of April 24, Beijing time, Nature, a top international academic journal, published online the progress of joint research made by Professor Luo Jingshan’s research group from the College of Electronic Information and Optical Engineering of Nankai University, and teams from the University of Cambridge and the Swiss Federal Institute of Technology in Lausanne (EPFL) in the field of photoelectrochemical (PEC) water splitting to produce hydrogen.

The study is entitled “High carrier mobility along the [111] orientation in Cu2O photoelectrodes”. Based on the solution electrochemical epitaxial growth technology, the team prepared three single-crystal cuprous oxide (Cu2O) films with different orientations, quantitatively analyzed the anisotropic photoelectric properties of Cu2O with femtosecond transient reflectance spectroscopy, and prepared polycrystalline Cu2O photoelectrodes with [111] as the primary crystal orientation based on the analysis conclusions, thereby making a breakthrough in PEC water splitting to produce hydrogen.
Reducing the cost of green hydrogen production holds the key to the rise of the hydrogen energy industry. PEC water splitting technology can directly convert intermittent solar energy into hydrogen energy. It is a promising renewable energy technology. As a natural p-type semiconductor, Cu2O is characterized by abundant raw material reserves, simple preparation methods, narrow band gap and suitable energy level position, making it the “star” material for efficient and inexpensive photoelectrode for hydrogen production.

In terms of improving the PEC performance of Cu2O, photon-generated carrier separation and transport efficiency enhancement are critical. There are few studies on the recombination process of carriers in the bulk phase of Cu2O.
In order to reveal the mechanism of influence of different crystal orientations on the carrier recombination in the bulk phase of Cu2O, Professor Jingshan Luo’s team from Nankai University, in collaboration with Professor Samuel D. Stranks’ team from the University of Cambridge and the team of Professor Michael Grätzel and Professor Anders Hagfeldt from the Swiss Federal Institute of Technology in Lausanne (EPFL), successfully prepared the single crystal Cu2O photoelectrode with [111], [110] and [100] crystal orientations using liquid-phase epitaxial growth technology.
Subsequently, the team analyzed the optoelectronic properties of Cu2O photoelectrodes with different crystal orientations. The results showed that the carrier mobility, conductivity, and carrier diffusion length of the single crystal Cu2O along the [111] crystal orientation were relatively better, demonstrating a relatively higher photocurrent density.
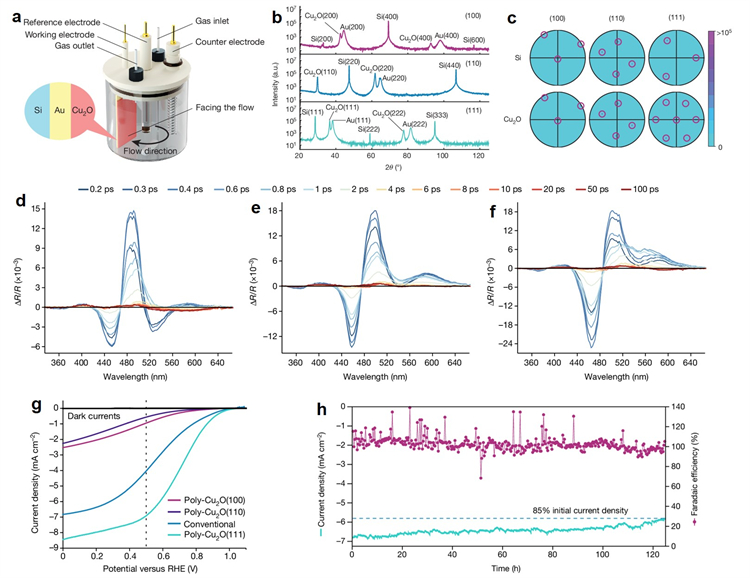
Fig. a, The electrochemical epitaxy set-up with the three-electrode configuration for liquid-phase epitaxial growth of Cu2O thin films. b, c, X-ray diffraction patterns (b) and pole figures generated from EBSD (c) for single-crystal epitaxial layers on Si substrates with reflections highlighted in circles; transient reflectance spectrum of 15 nm single-crystal Cu2O film with (100) (d), (110) (e) and (111) (f) crystal orientations. g The current density-voltage response curves of traditional polycrystalline Cu2O and [111] orientation-enhanced polycrystalline Cu2O photocathode [poly-Cu2O (111)] under simulated illumination of solar AM1.5 G. h. Stability test on poly-Cu2O (111) at the fixed bias of 0.5 V (versus the RHE) and Faradaic efficiency for the hydrogen evolution reaction.
Based on the analysis results, the team prepared a poly-Cu2O photocathodes with an extraordinarily pure [111] orientation, demonstrating the advantages of the electronic properties along the [111] orientation. The current density at 0.5 V versus the RHE reaches 7 mA cm-2 ( a 75% improvement compared with the state-of-the-art electrodeposited devices).
Moreover, the team explored the effects of different crystal orientations and corresponding exposure terminating facets on the stability of the Cu2O photoelectrode, and the results revealed that the [111] crystal orientation and extensive exposure of (111) facets ensure more excellent stability for Cu2O photocathode.

It is reported that this study innovatively developed the technology of preparing single-crystal Cu2O thin films using liquid-phase epitaxial growth, quantitatively analyzed the optoelectronic properties of Cu2O films with different crystal orientations, and revealed the influence of different crystal orientations on bulk phase carrier recombination.
Based on this finding, the team enhanced the [111] crystal orientation of the polycrystalline Cu2O photoelectrode to increase the photocatalytic performance of the planar Cu2O photocathode. These findings provide a widely applicable strategy for the improvement of oxides in the fields of photovoltaics, transistors, detectors, and solar fuels.

Professor Jingshan Luo’s team has been focusing on improving the performance of Cu2O photocathode for hydrogen production for many years. A nanostructure strategy was used to achieve 10 mA cm-2 for the saturation photocurrent density of the Cu2O photocathode for the first time (Nano Letters, 2016, 16, 1848-1857). Then, a novel Ga2O3 electron transfer layer was introduced to increase the onset potential of the Cu2O photocathode for hydrogen production to the recorded value of 1 V (vs. RHE) (Nature Catalysis, 2018, 1, 412-420). Degradation mechanism of Cu2O photoelectrodes was also systematically studied (ACS Appl. Mater. Interfaces, 2021, 13, 46, 55080–55091). Recently, the photovoltage of Cu2O photoelectrode was further increased to 1.07 V based on a dual buffer layers strategy (Nature Communications, 2023, 14, 7228).
Nankai University is one of the corresponding organizations for this work, and Professor Jingshan Luo from the College of Electronic Information and Optical Engineering is the co-corresponding author of the paper.
URL:
https://www.nature.com/articles/s41586-024-07273-8
(Edited and translated by Nankai News Team.)









